How Multiple Sclerosis Is Diagnosed: Tests, MRI, and What to Expect
What you will learn in this post
Getting a diagnosis of multiple sclerosis is rarely a single moment. It is a process — sometimes a long one — involving several different tests, multiple appointments, and a set of criteria that neurologists must satisfy before they can say with confidence that MS is what they are dealing with. This post walks you through every step of that process in plain language: what each test involves, what it is looking for, what the results mean, and why the whole journey can take longer than most people expect.
Quick answer: Multiple sclerosis is diagnosed through a combination of MRI scanning, neurological examination, cerebrospinal fluid analysis (lumbar puncture), and evoked potential tests. No single test confirms MS on its own. Neurologists use the McDonald criteria — a set of evidence-based rules requiring proof of damage in multiple locations in the central nervous system, occurring at more than one point in time — to make a formal diagnosis. This process can take months or, in some cases, years.
At a glance: the main tests used to diagnose MS
| Test | What it looks for | Always required? | What a positive result suggests |
|---|---|---|---|
| MRI of brain and spinal cord | Lesions (areas of myelin damage) | Yes — the primary tool | Demyelination consistent with MS |
| Lumbar puncture (spinal tap) | Oligoclonal bands in cerebrospinal fluid | Often, but not always | Inflammation in the central nervous system |
| Evoked potential tests | Slowed nerve signal transmission | Sometimes | Subclinical nerve damage not visible on MRI |
| Blood tests | Conditions that can mimic MS | Yes — to rule out alternatives | Vitamin deficiencies, lupus, Lyme disease, and others |
| Neurological examination | Reflexes, coordination, vision, sensation | Yes — at every appointment | Clinical evidence of nervous system involvement |
What does diagnosing MS actually involve?
If you have been referred to a neurologist with symptoms that might be MS — tingling in the limbs, an episode of blurred vision, unusual fatigue, balance problems that came on suddenly — the first thing to understand is that the diagnostic process is deliberately thorough. That thoroughness is not bureaucratic. It exists because MS shares symptoms with a surprising number of other conditions, because the consequences of a misdiagnosis in either direction are significant, and because the criteria neurologists use to confirm MS require evidence that simply takes time to gather.
The process typically begins with a detailed neurological examination. Your neurologist will assess your reflexes, eye movements, coordination, sensation, and muscle strength. This is not a formality — it is clinically essential. The findings from the examination guide which tests are ordered and how the results are interpreted.
From there, the workup almost always includes an MRI of the brain and usually the spinal cord, blood tests to exclude conditions that can look like MS, and frequently a lumbar puncture. Evoked potential tests may be added depending on what the MRI and examination show. The results of all of these are then assessed against the McDonald criteria — the internationally accepted framework for diagnosing MS — before a diagnosis is made or, in some cases, before a period of monitoring begins.
Why MS is hard to diagnose — and why it takes time
One of the most common and most painful experiences in the MS journey is the gap between the first symptom and a confirmed diagnosis. Research has consistently found that the average time from first symptom to diagnosis ranges from several months to several years, and for some people it is longer still.
This delay is not a failure of medicine. It reflects several genuine challenges that make early MS genuinely difficult to identify with certainty.
The first is that early MS symptoms are frequently episodic. They appear, cause concern, and then partially or fully resolve — which can lead both patients and doctors to attribute them to stress, a virus, a trapped nerve, or fatigue. A single episode of blurred vision that clears in two weeks is easy to dismiss. It is only in retrospect, when a second episode occurs months later, that the pattern becomes clinically visible.
The second is that many MS symptoms are non-specific. Tingling, fatigue, and balance problems are caused by dozens of conditions. The neurological system is not generous with unique fingerprints — similar symptoms can arise from very different underlying causes. Ruling out those other causes takes time and testing.
The third is that the McDonald criteria — by design — require evidence of damage in at least two separate locations in the central nervous system (called dissemination in space) and evidence that this damage occurred at more than one point in time (called dissemination in time). If your first MRI shows one lesion and no previous symptoms can be documented, you may not yet meet the criteria, even if MS is strongly suspected. A second MRI three to six months later showing a new lesion would satisfy dissemination in time — but that wait is inherently part of the process.
This is one of the hardest things to communicate to someone in the middle of it. The wait is not negligence. It is, in many cases, medically necessary.
The McDonald criteria: how neurologists decide
The McDonald criteria are the internationally accepted diagnostic framework for MS, last updated in 2017. They are named after the neurologist W. Ian McDonald and exist to standardise the diagnosis across different clinical settings and ensure that MS is only diagnosed when there is sufficient neurological evidence to support it.
The two core requirements of the McDonald criteria are:
- Dissemination in space means that MRI or clinical evidence must show damage in at least two distinct areas of the central nervous system — for example, both the brain and the spinal cord, or two different regions of the brain. This distinguishes MS from conditions that affect only one location.
- Dissemination in time means that there must be evidence that neurological damage occurred at more than one point in time. This can be demonstrated by two separate clinical attacks (relapses) separated by at least 30 days, or by MRI showing both old and new lesions, or by a new lesion appearing on a follow-up MRI compared to an earlier one.
The 2017 revision of the criteria also allowed cerebrospinal fluid analysis — specifically the presence of oligoclonal bands — to substitute for some requirements around dissemination in time, which has helped accelerate diagnosis in some cases where waiting for a second MRI would otherwise delay treatment.
Understanding these criteria matters not just because they explain the diagnosis, but because they explain the timeline. Your neurologist is not being slow or cautious without reason. They are building the specific body of evidence the criteria require.
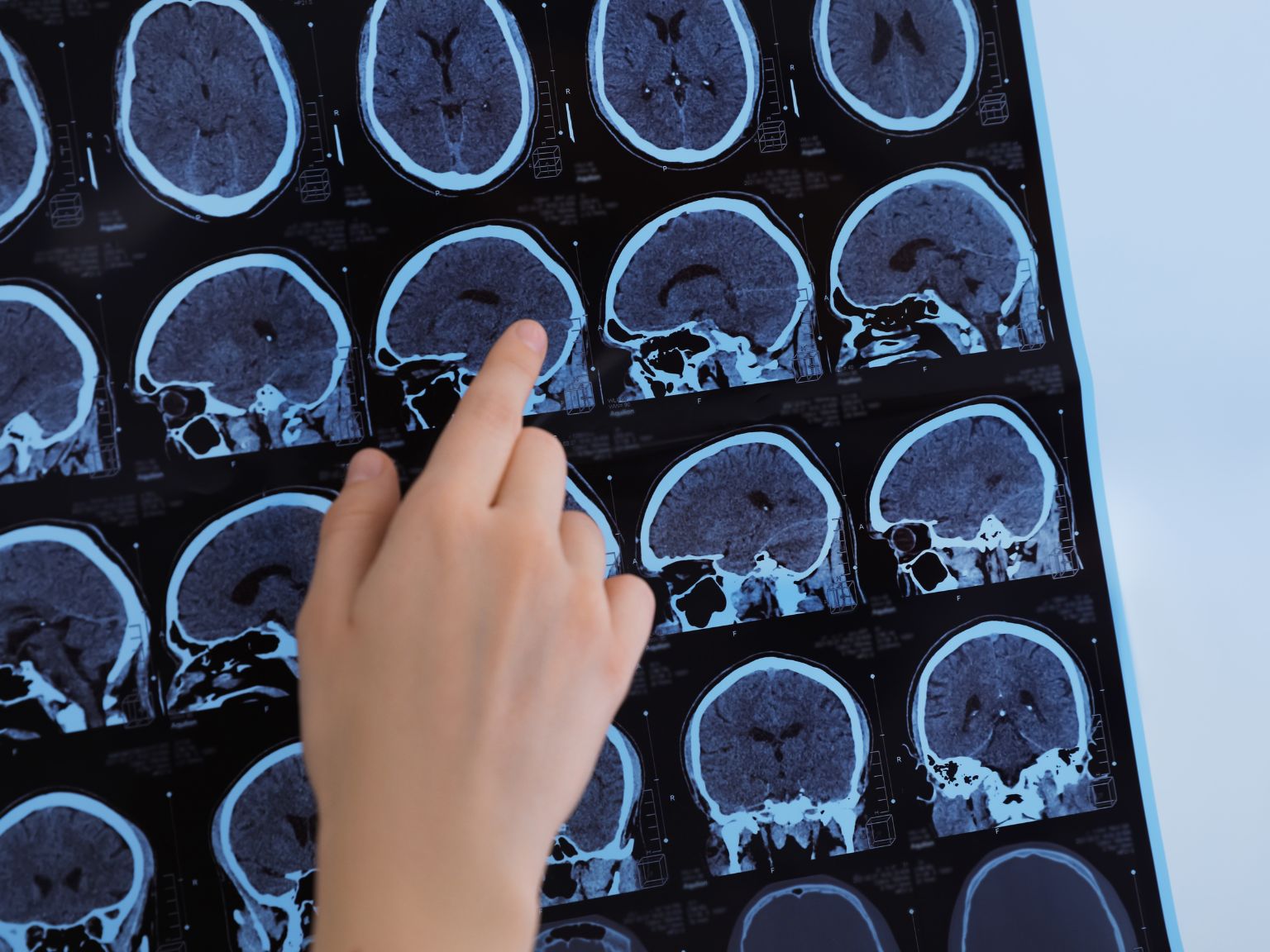
MRI scanning for MS: what it shows and what it means
MRI — magnetic resonance imaging — is the most important single tool in the MS diagnostic process. It is non-invasive, does not use radiation, and is highly sensitive to the kind of damage MS causes in the brain and spinal cord.
When myelin — the protective sheath around nerve fibres — is damaged by the immune system, the affected areas become visible on MRI as lesions: bright spots or dark spots depending on the imaging sequence being used. In MS these lesions have characteristic appearances, sizes, and locations that help distinguish them from the lesions caused by other conditions.
A standard MS MRI will typically image the brain, and often the cervical spinal cord (the portion of the spinal cord in the neck), using several different sequences. The two most important for MS diagnosis are T2-weighted imaging and T1-weighted imaging with gadolinium contrast.
T2-weighted images show the total burden of lesions — both old and new. Active areas of inflammation and older, inactive scarring both appear bright on T2. This gives the neurologist a picture of how much cumulative damage has occurred.
T1-weighted images with gadolinium contrast show only active lesions — areas where the blood-brain barrier is currently disrupted because inflammation is ongoing. Gadolinium is a contrast agent injected into a vein before the scan that leaks into areas of active inflammation and makes them visible. An enhancing lesion on a gadolinium-enhanced T1 scan is evidence of current disease activity, which is clinically important both for diagnosis and for monitoring treatment response.
It is important to understand that a single MRI cannot confirm MS on its own. What an MRI can do is show lesions consistent with MS in locations typical for the condition — and those findings, combined with clinical history and other test results, build the picture the McDonald criteria require.
Equally, a normal MRI does not rule out MS, particularly early in the disease. In some people, especially those with spinal cord involvement rather than brain involvement, early MRIs can appear near-normal while clinical symptoms are already present. This is why the full diagnostic workup matters, and why your neurologist will not rely on imaging alone.
What MS lesions look like on an MRI
MS lesions in the brain are typically ovoid (oval-shaped), and have a strong predilection for certain locations: the periventricular white matter (the tissue adjacent to the fluid-filled ventricles of the brain), the juxtacortical region (just beneath the outer surface of the brain), the infratentorial region (the brainstem and cerebellum), and the spinal cord. This pattern of distribution — multiple lesions in these specific areas — is one of the features that makes MS lesions recognisable.
Neurologists use a set of criteria called the MAGNIMS or Barkhof criteria to assess whether brain MRI lesions are typical of MS based on their number, size, shape, and location. An MRI report that describes "lesions characteristic of demyelinating disease" in the right locations carries significant diagnostic weight.
Will one MRI confirm MS?
Usually not. A single MRI scan can provide strong supportive evidence for MS — it can demonstrate dissemination in space if lesions appear in multiple characteristic locations — but confirming dissemination in time typically requires either a follow-up MRI showing new lesions, or a combination of the initial MRI findings with cerebrospinal fluid analysis.
There are exceptions. The 2017 McDonald criteria allow a diagnosis of MS to be made on the basis of a single MRI if that MRI shows both gadolinium-enhancing and non-enhancing lesions, since these represent damage occurring at different time points. But this is not always the case, and for many people a follow-up MRI three to six months later will be part of the diagnostic process.
Lumbar puncture (spinal tap): what happens and why
A lumbar puncture — sometimes called a spinal tap — is a procedure in which a small amount of cerebrospinal fluid (CSF) is withdrawn from the space around the lower spinal cord using a thin needle inserted into the lower back. It is performed under local anaesthetic, usually takes 20 to 30 minutes, and is followed by a period of rest.
The cerebrospinal fluid sample is then analysed in a laboratory. In the context of MS diagnosis, the key finding being looked for is oligoclonal bands — distinctive proteins produced by immune cells that have entered the central nervous system and are generating an inflammatory response. Oligoclonal bands are found in the CSF of approximately 85 to 90 percent of people with MS.
It is important to understand what the presence of oligoclonal bands does and does not mean. Their presence confirms that there is inflammation in the central nervous system — but it does not on its own confirm MS, because other neurological conditions can also produce them. Their absence, conversely, makes MS less likely but does not rule it out.
Under the 2017 McDonald criteria, the presence of oligoclonal bands in the CSF can substitute for certain requirements around dissemination in time, meaning that in some cases a positive CSF result can shorten the diagnostic process by removing the need to wait for a second demyelinating event.
Is a lumbar puncture always required to diagnose MS?
No. A lumbar puncture is not mandatory in every case. In patients whose MRI findings and clinical history clearly satisfy all the McDonald criteria requirements for dissemination in space and time, a diagnosis can be made without CSF analysis. However, most neurologists will recommend a lumbar puncture as part of the workup because the CSF findings add meaningful diagnostic certainty and can help distinguish MS from conditions that can look similar on MRI.
If you have been told you need a lumbar puncture and are anxious about the procedure, it is worth knowing that serious complications are uncommon. The most frequent side effect is a post-procedure headache, which occurs in roughly 10 to 20 percent of people and usually resolves within a day or two with rest and fluids.
Evoked potential tests: measuring nerve signal speed
Evoked potential tests measure how quickly and reliably the brain receives and processes signals from the eyes, ears, or skin. In MS, demyelination slows down nerve signal transmission — sometimes in ways that are clinically silent, meaning the person has no symptoms in that pathway. Evoked potential tests can detect this slowing even when it is not causing noticeable problems, and that detection can provide additional diagnostic evidence.
The three types most commonly used in MS diagnosis are:
- Visual evoked potentials (VEPs) measure the brain's electrical response to a flickering checkerboard pattern viewed with one eye at a time. MS frequently causes inflammation of the optic nerve (optic neuritis), which slows the VEP signal even after the visual symptoms have resolved. A delayed or abnormal VEP in someone whose vision appears normal is evidence of previous optic nerve demyelination — and therefore evidence of dissemination in space and potentially time.
- Somatosensory evoked potentials (SSEPs) measure how quickly sensory signals travel from the limbs through the spinal cord to the brain. They can detect demyelination in sensory pathways in the spinal cord.
- Brainstem auditory evoked potentials (BAEPs) measure the brain's response to sound and can detect demyelination in the brainstem auditory pathways.
Evoked potential tests are most useful diagnostically when the clinical picture is incomplete — when symptoms are present but the MRI does not yet show enough lesions to satisfy dissemination in space, for example, or when the neurologist is trying to identify silent lesions to support the criteria.
Blood tests and what they rule out
Blood tests are not used to diagnose MS directly — there is currently no blood biomarker that confirms MS. Their role in the diagnostic workup is to exclude other conditions that can produce neurological symptoms similar to those of MS.
The conditions most commonly screened for include vitamin B12 deficiency, which can cause spinal cord damage and peripheral neuropathy; neuromyelitis optica spectrum disorder (NMOSD), which is sometimes confused with MS but requires different treatment and is identified by a specific antibody called AQP4-IgG; lupus and other autoimmune conditions; Lyme disease, particularly in geographic areas where it is prevalent; thyroid dysfunction; and HIV.
Some of these conditions are clinically important to exclude not just for diagnostic accuracy but because the treatments used for MS — particularly disease-modifying therapies that modulate the immune system — could cause serious harm if used in someone who actually has one of the mimicking conditions.
If your blood test results come back normal, that is an important piece of the diagnostic picture. Normal blood tests, combined with abnormal MRI findings and clinical symptoms, direct the focus firmly toward MS and away from the conditions the blood work was designed to catch.
Conditions that mimic MS
Understanding what MS can be confused with is genuinely useful for people in the diagnostic process, because it explains both why the workup is thorough and why some tests that seem unrelated to the brain are ordered.
- Neuromyelitis optica spectrum disorder (NMOSD) is the most clinically important MS mimic. It causes similar symptoms — optic neuritis, spinal cord attacks — but is a different disease driven by a different immune mechanism, and is identified by the AQP4-IgG antibody in the blood. Treating NMOSD as if it were MS can worsen the condition.
- Migraine with aura produces white matter lesions on MRI that can resemble MS lesions and is a common source of diagnostic uncertainty, particularly in younger women.
- Antiphospholipid syndrome, lupus, and other connective tissue diseases can cause neurological symptoms and white matter changes.
- Vitamin B12 deficiency causes subacute combined degeneration of the spinal cord — a condition that can produce weakness, sensory loss, and balance problems very similar to MS.
- Lyme neuroborreliosis, in regions where Lyme disease is prevalent, can produce neurological symptoms and MRI changes that overlap with MS.
The process of ruling these out is not a delay to the real diagnosis. It is the diagnosis — the systematic narrowing down of possibilities until what remains is either MS or something that needs its own treatment.
What to expect at your neurology appointment
If you are attending your first neurology appointment with suspected MS, knowing what to expect can reduce the anxiety that comes from the unknown.
The appointment will almost certainly begin with a detailed history. Your neurologist will ask about your current symptoms, when they started, whether they have changed or improved, and whether you have had any previous episodes of neurological symptoms that resolved — sometimes years earlier. That last question matters enormously, because previous episodes that were attributed to other causes may, in retrospect, represent early MS activity.
The neurological examination that follows typically takes 20 to 40 minutes and assesses eye movements and pupillary reflexes, visual acuity, facial sensation, swallowing and speech, upper and lower limb strength, reflexes, coordination (including finger-to-nose and heel-to-shin testing), gait and balance, and sensation including vibration sense and proprioception.
After the examination your neurologist will discuss their findings and explain which tests they are recommending. In many cases the MRI will already have been ordered before the appointment — but even if not, the examination findings will determine what is requested.
Questions to bring, records to prepare
The most useful thing you can do before a neurology appointment is write down a timeline of symptoms. Include the first time you noticed anything unusual, even if it seemed minor or resolved quickly. Include anything that might now look like an early episode in retrospect. Your neurologist is building a clinical history — the more complete that history is, the more useful the appointment will be.
Bring any previous MRI scans, even of a different body part. Bring any blood test results from the past two years. If you have seen any other specialist — an ophthalmologist for visual symptoms, a physiotherapist for balance, a GP for fatigue — bring those letters or results if you have them.
The questions most worth asking at the appointment include: What do you think is causing my symptoms? What tests are you recommending and what are they looking for? What will happen if the tests are inconclusive? How long is the diagnostic process likely to take? Who should I contact if my symptoms worsen before my next appointment?
You do not need to have all of this memorised. Writing the questions down and bringing them to the appointment is entirely appropriate — and a good neurologist will expect it.
After diagnosis: what happens next
A confirmed MS diagnosis is not the end of the process. It is the beginning of a different one.
The first priority after diagnosis is connecting with an MS specialist and, where available, an MS specialist nurse. These are not the same as a general neurologist. MS nurses in particular are among the most valuable clinical relationships a newly diagnosed person can have — they are more accessible than consultants, understand the practical day-to-day questions that rarely make it into clinic appointments, and can often navigate the next steps more quickly.
The second priority is a conversation about disease-modifying therapies (DMTs). These are medications that reduce the frequency and severity of relapses and slow the accumulation of disability. They do not cure MS and they do not repair existing damage — but starting an appropriate DMT early, where clinically indicated, is one of the most important decisions of the early post-diagnosis period. The specific DMT recommended will depend on your MS type, the activity visible on your MRI, and your personal circumstances and preferences.
The third is emotional support. A new diagnosis of MS is a significant life event, and the range of reactions — shock, relief that there is finally an answer, grief, fear, and eventually a kind of determined adaptation — is wide and entirely valid. Connecting with a peer support group, a counsellor experienced with chronic illness, or an online MS community in the early period can make a meaningful difference to how you navigate the months ahead.
If you are reading this before a diagnosis has been confirmed, knowing this is what follows may make the process feel less like a cliff edge and more like a door — one that leads to a path that, while not the one you would have chosen, is navigable.
Frequently asked questions about the MS diagnosis process
How long does it take to get an MS diagnosis?
There is no fixed timeline. Some people receive a diagnosis within weeks of their first appointment, particularly if they present with a clear clinical picture and MRI findings that satisfy the McDonald criteria without further testing. For others — especially those whose symptoms were episodic and resolved before they sought help, or whose initial MRI was not clearly diagnostic — the process can take one to three years or longer. The average across published studies is approximately two to three years from first symptom to confirmed diagnosis, though this varies significantly by country and healthcare system.
Can MS be diagnosed from an MRI alone?
Not definitively, though MRI is the most important single tool in the process. An MRI that shows lesions in characteristic locations consistent with demyelination is strong evidence for MS — but the diagnosis requires the full McDonald criteria to be satisfied, which typically involves clinical history and often CSF analysis in addition to imaging.
What does it mean if my MRI shows white matter lesions but I haven't been diagnosed with MS?
White matter lesions on an MRI are not specific to MS. They can be caused by migraine, small vessel disease, normal aging, hypertension, and a range of other conditions. If your MRI shows white matter changes but your clinical history and other test results do not satisfy the McDonald criteria for MS, it is entirely possible that another explanation is more likely. This is one of the reasons MRI is not used in isolation.
Is a lumbar puncture painful?
The procedure itself is performed under local anaesthetic, which means the actual needle insertion is usually felt as pressure rather than pain. The most common aftereffect is a post-procedure headache caused by a small leak of cerebrospinal fluid from the puncture site. This affects roughly one in ten to one in five patients, typically begins within 24 hours, and usually resolves with rest and hydration. Lying flat for a few hours after the procedure reduces the risk.
Can MS be ruled out if all my tests are normal?
A normal MRI and normal CSF analysis significantly reduce the likelihood of MS, but do not definitively rule it out in all cases. In very early disease, or in MS subtypes with predominantly spinal cord involvement, early MRI scans can appear normal or near-normal. If symptoms continue or recur, further investigation over time remains appropriate.
What is a clinically isolated syndrome?
A clinically isolated syndrome (CIS) is a first episode of neurological symptoms caused by inflammation and demyelination in the central nervous system — the kind of episode that could be the first presentation of MS, but where the full McDonald criteria for MS have not yet been satisfied. People with CIS are monitored closely, and in some cases DMT treatment is recommended even before a formal MS diagnosis, to reduce the risk of a second episode that would confirm MS.
Does a normal neurological examination mean I don't have MS?
Not necessarily. MS can be present and produce significant MRI changes even when the neurological examination is clinically normal, particularly in early disease. Conversely, an abnormal neurological examination is an important positive finding when present. The examination is one component of the diagnostic picture, not a standalone test.
Why do I need blood tests if my symptoms are neurological?
Because several conditions that affect the blood and immune system produce neurological symptoms that overlap significantly with MS. Excluding those conditions — particularly neuromyelitis optica, B12 deficiency, lupus, and Lyme disease — is a necessary part of the diagnostic process, not a detour away from it.
Can acupuncture help during the MS diagnostic process or after diagnosis?
Many people experiencing neurological symptoms — whether or not a diagnosis has been confirmed — seek complementary support alongside their conventional medical workup. Acupuncture is used by some people with MS and neurological conditions to support symptom management, particularly for pain, fatigue, and stress during what is often a prolonged and anxiety-provoking diagnostic period. It is not a treatment for MS itself, but as a supportive therapy used alongside neurological care, many patients find it a valuable part of managing their overall wellbeing. If you are currently going through the diagnostic process and want to discuss whether acupuncture might support you, our team at Dr Tang Acupuncture Clinic is here to talk through what that might look like for your situation.
Conclusion
The diagnosis of multiple sclerosis is not based on a single test or a single moment. It is a careful, step-by-step process that combines neurological examination, MRI imaging, blood tests, and sometimes cerebrospinal fluid analysis. Each part of this process helps neurologists rule out other conditions and confirm whether the pattern of nerve damage matches MS.
While the waiting period for a diagnosis can feel uncertain and emotionally challenging, this thorough approach is important to ensure accuracy. MS shares symptoms with many other neurological conditions, so time and detailed evaluation are essential before reaching a final conclusion.
For individuals experiencing ongoing neurological symptoms or seeking further clarity during this process, supportive care options are also considered by many patients in Dubai. Clinics such as Dr Tang Acupuncture Clinic provide complementary approaches alongside medical evaluation, focusing on overall nervous system support, symptom relief, and general well-being.
If symptoms persist or evolve, early consultation with a qualified specialist is always recommended. With proper diagnosis, timely monitoring, and the right care plan, patients can move forward with clarity and a better understanding of their condition.
